 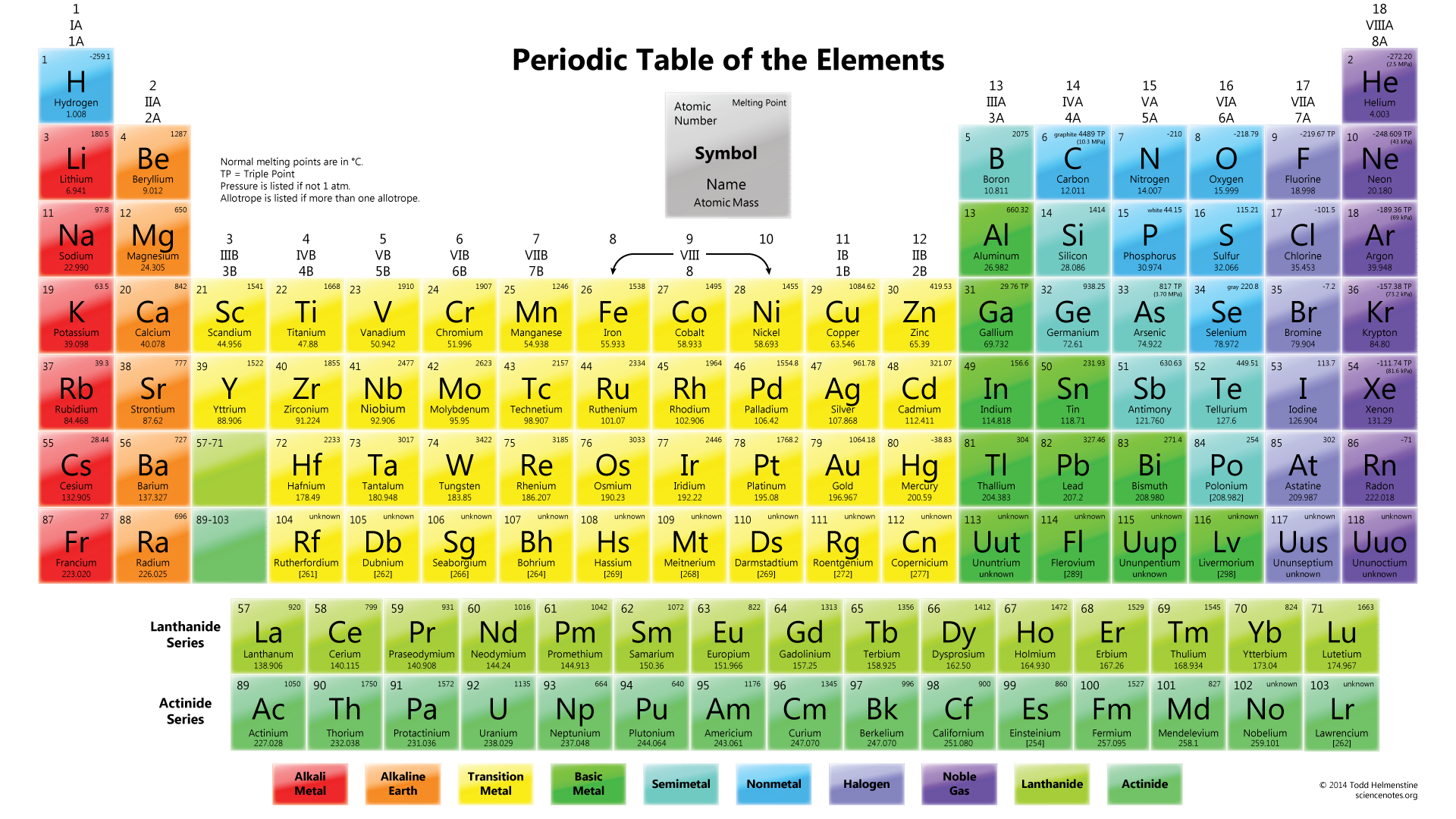
Just two strategies existed at the time to categorize these elements: separating them into metals and nonmetals or grouping them by an element's number of valence electrons (or those electrons in the outermost shell). At the time, there were 63 known chemical elements, each with an atomic weight calculated using Avogadro's hypothesis, which states that equal volumes of gases, when kept at the same temperature and pressure, hold the same number of molecules. Putting the elements in any kind of order would prove quite difficult. (Image credit: Oxford Science Archive/Print Collector/Getty Images) For instance, all the group 18 elements are inert gases, meaning they don't react with any other elements. Elements that occupy the same column on the periodic table (called a "group") have identical valence electron configurations and consequently behave in a similar fashion chemically. As an example, elements in Group 8A (or VIIIA) all have a full set of eight electrons in the highest-energy orbital, according to chemist William Reusch, on his webpage at Michigan State University. The columns, or groups, on the periodic table represent the atomic elements that have the same number of valence electrons, or those electrons in the outermost orbital shell. (Atoms have protons and neutrons in their nucleus, and surrounding that, they have their electrons arranged in orbitals, where an atomic orbital is a math term that describes the location of an electron as well as its wave-like behavior.)įor instance, period 1 includes elements that have one atomic orbital where electrons spin period 2 has two atomic orbitals, period 3 has three and so on up to period 7. Magnesia, a district of Eastern Thessaly in GreeceĪlumina, from Latin alumen (gen.The horizontal rows on the periodic table are called periods, where each period number indicates the number of orbitals for the elements in that row, according to Los Alamos National Laboratory. Symbol Na is derived from Neo-Latin natrium, coined from German Natron, ' natron'. 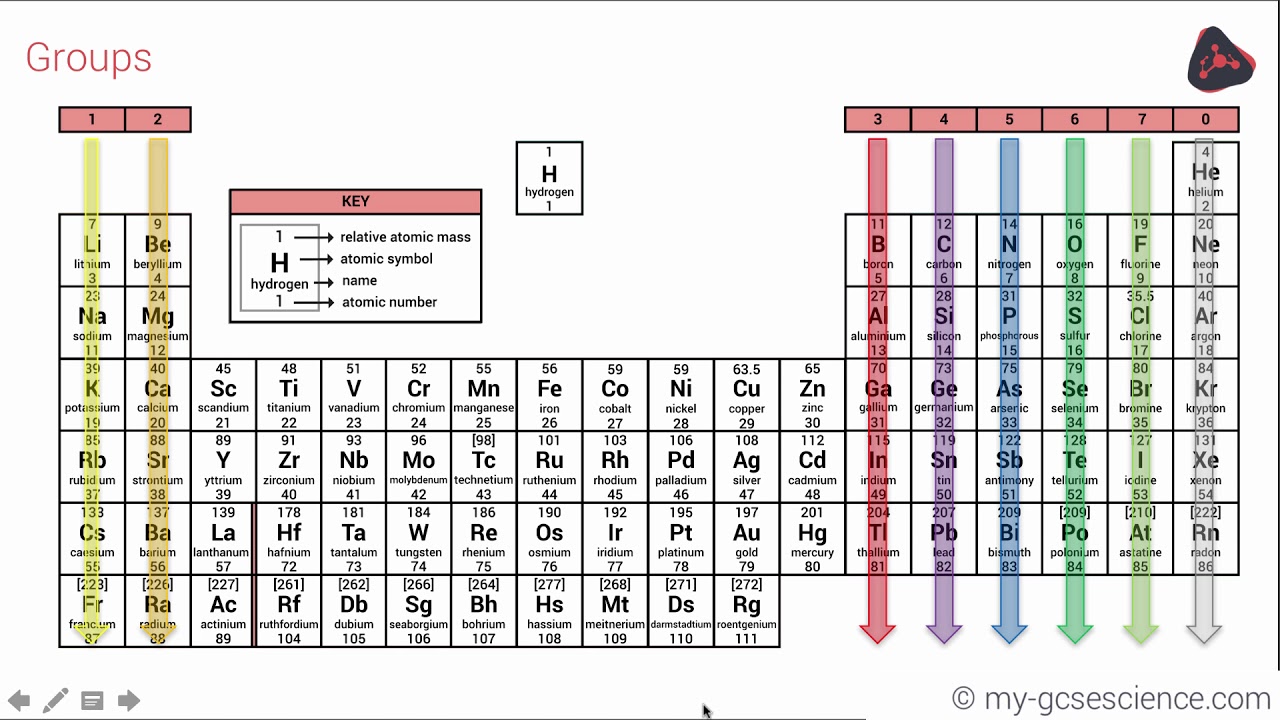
Greek elements hydro- and -gen, ' water-forming'īeryl, a mineral (ultimately from the name of Belur in southern India) īorax, a mineral (from Arabic bawraq, Middle Persian * bōrag) For more detailed information about the origins of element names, see List of chemical element name etymologies. Like the periodic table, the list below organizes the elements by the number of protons in their atoms it can also be organized by other properties, such as atomic weight, density, and electronegativity. It is a tabular arrangement of the elements by their chemical properties that usually uses abbreviated chemical symbols in place of full element names, but the linear list format presented here is also useful. The definitive visualisation of all 118 elements is the periodic table of the elements, whose history along the principles of the periodic law was one of the founding developments of modern chemistry. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z). This is a list of the 118 chemical elements that have been identified as of 2023. List of the 118 identified chemical elements 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
